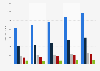
U.S. NDA/BLA filing approval for new drugs in select disease areas 2006-2015
This statistic shows the percent approval for NDA/BLA filings for new drugs in the U.S., within the period from January 1, 2006 to December 31, 2015, by disease area. It was found that 79 percent of oncology drugs were approved by the FDA on first review, compared to just 37 percent of psychiatry drugs.