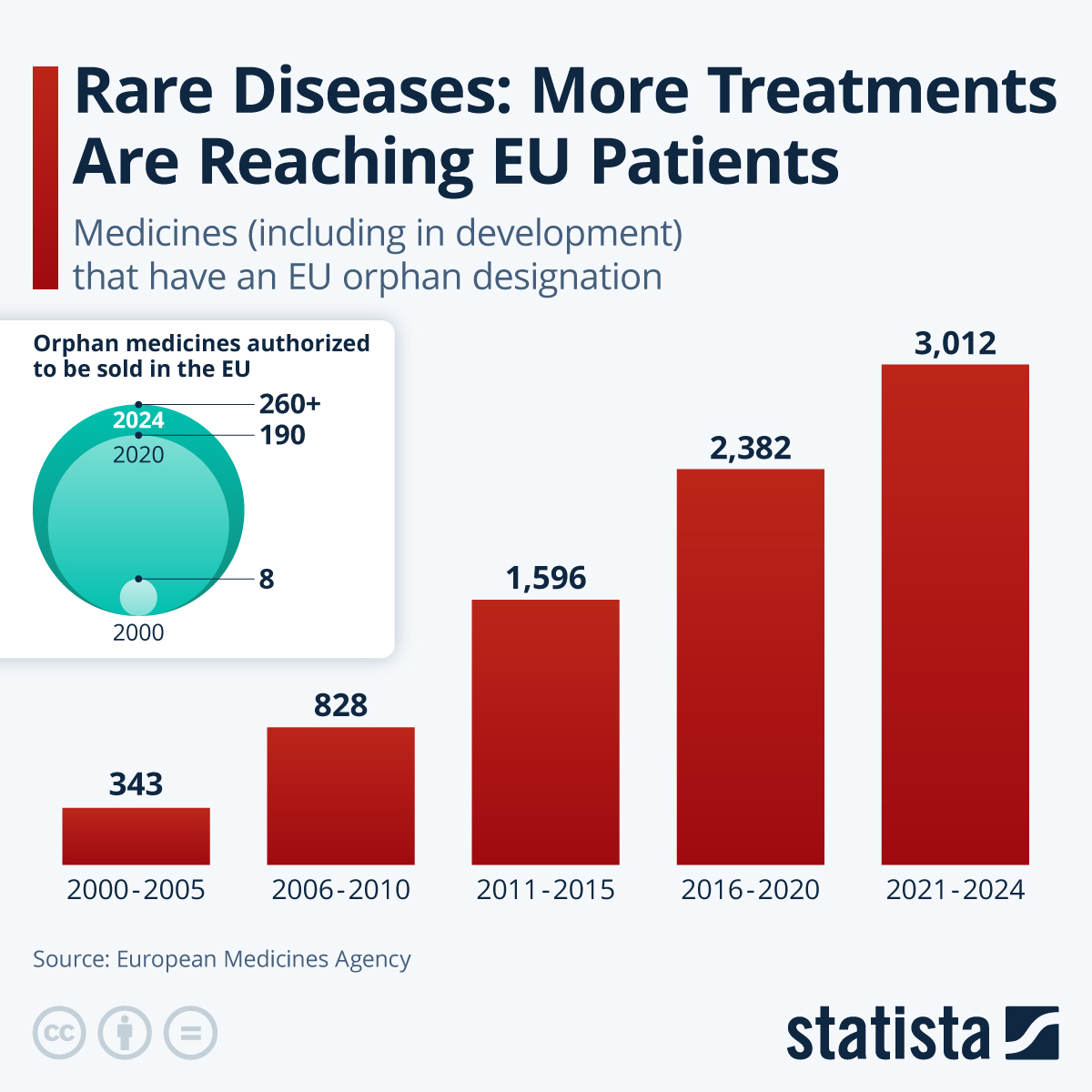
Rare-disease research has long struggled with relatively small patient populations and limited commercial incentives. But in the European Union, the orphan-drug framework introduced in 2000 has significantly contributed to the development of medicines for rare diseases, by offering incentives such as fee reductions, market exclusivity and protocol assistance to pharmaceutical companies.
According to figures compiled by the European Medicines Agency, the number of medicines that have received an EU orphan designation (early regulatory status before market authorization, including many still in development), has climbed steadily over the past two decades, reaching 3,012 in 2024. At the same time, the number of orphan medicines finally authorized for sale in the EU has also sharply risen, from just eight in 2000 to 190 by 2020, and more than 260 by 2024 - a sign that more projects are making it from development into marketed therapies recently.