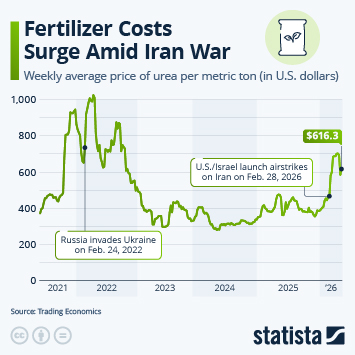
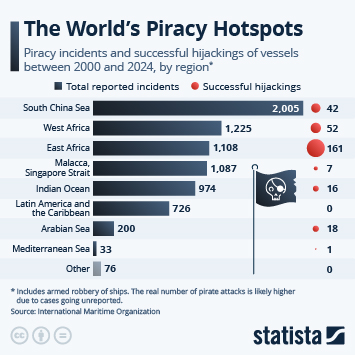
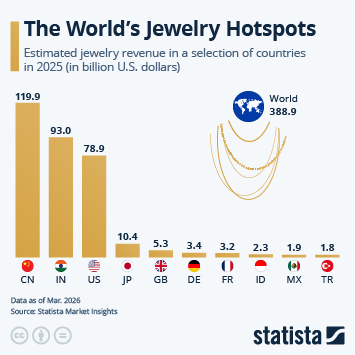
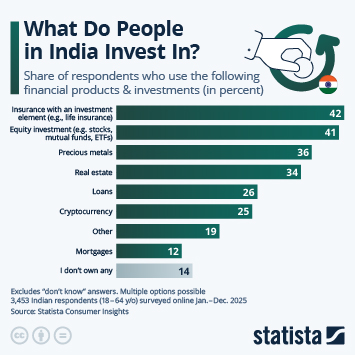
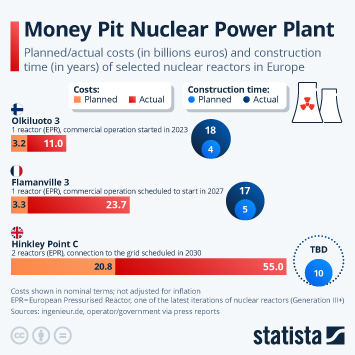
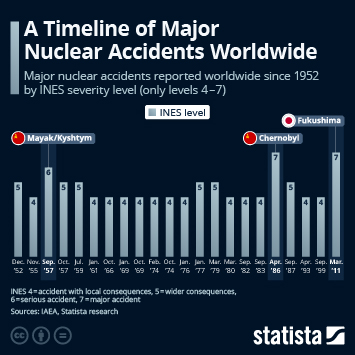
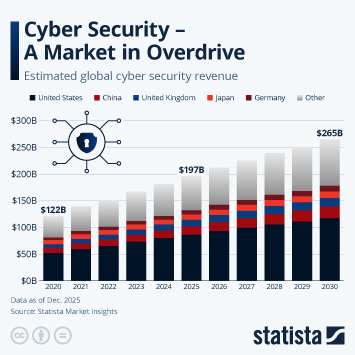
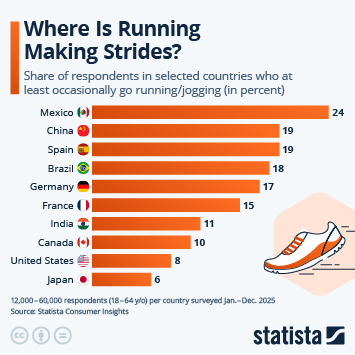
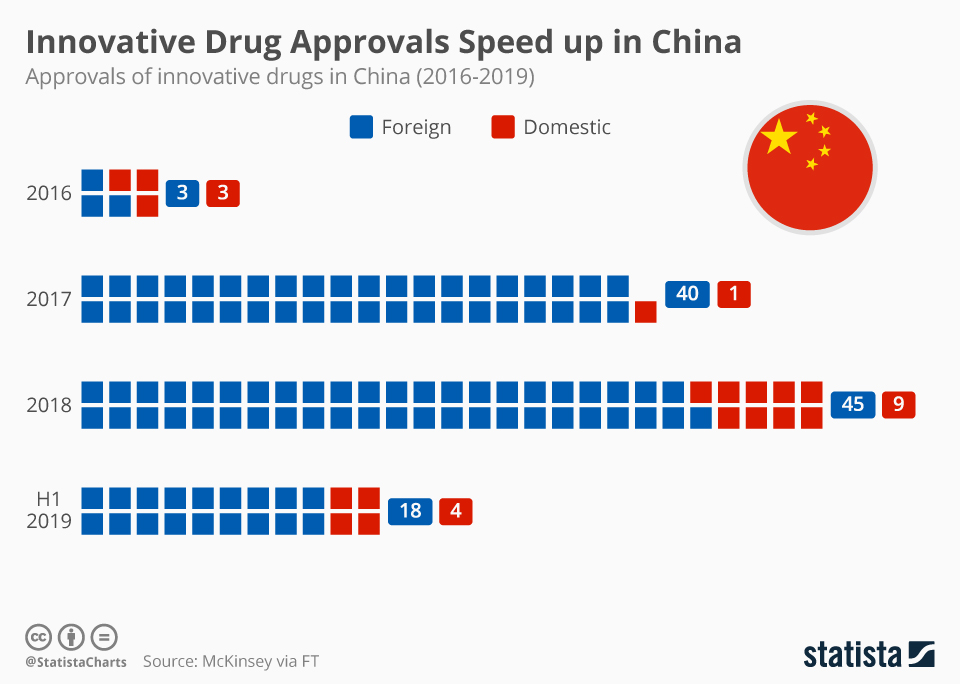
Regulatory changes have paved the way in China for faster approval of innovative drugs. Approval numbers have been rising fast since 2017.
The Chinese pharma sector had been lobbying the government for some years to create a faster approval process, which was overhauled in 2015. The old one had previously caused a time lag in the approval of new, innovative drugs of five to eight years behind other countries, according to Deloitte.
Now, 2018 numbers suggest that China’s approval agency, the China Food and Drug Administration (CFDA), is almost en par with its U.S. counterpart, the Food and Drug Administration (FDA). China has also already beaten some other countries when it comes to innovative drug approval. In December, anemia drug Roxadustat was allowed to enter the Chinese market before any other – a first for China.
Approval of new drugs in China is now possible in around three years. For drugs meeting criteria of urgent need, fast-track application were granted in only 16 months in some cases. This is way below the FDA average of 12 years.
Despite the gains in approvals, few of the newly legal drugs have been covered by China’s public insurance program, leaving some companies unsure about the benefits of approval in China since this might leave treatments out of financial reach for many Chinese.
Drug Approvals
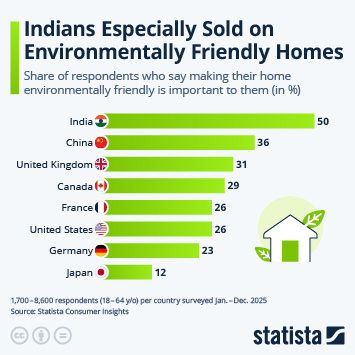
Innovative Drug Approvals Speed up in China
Description
This chart shows the number of foreign and domestic innovative drug approvals in China between 2016 and 2019.